Profiling with CourseMap AI
Discover how we profile trial participants and boost trial power
CourseMap AI: how it works
CourseMap AI is designed to translate advanced disease progression modeling into a workflow that is compatible with real clinical trial constraints. It combines scientifically validated digital twin models with rigorous operational processes.
Generating individual long-term disease trajectories
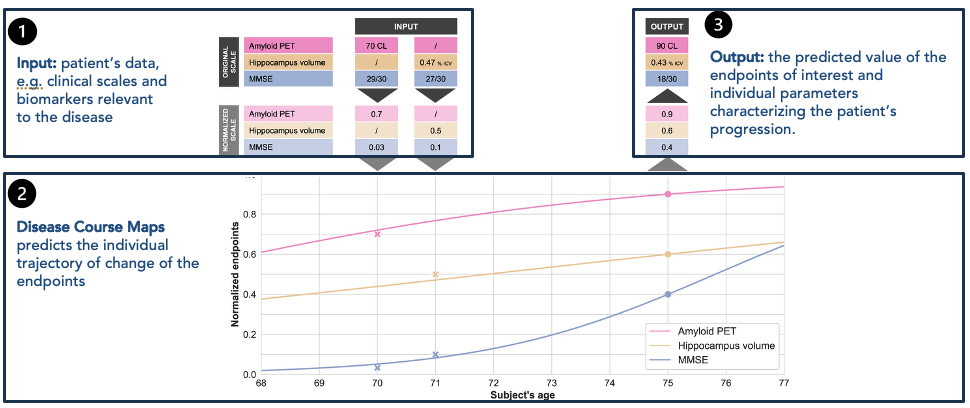
At the core of CourseMap AI is a digital twin approach to disease progression modeling. For each participant, the technology constructs an individualized representation of disease trajectory by integrating baseline clinical, cognitive, imaging, and biological data.
These digital twins capture both population-level structure and inter-individual variability, allowing long-term disease trajectories to be inferred even when only few observations are available, even at only one time-point.
Patented generative AI technology
Models available for several neurodegenerative diseases
Proven best performance in predicting cognitive decline
Scientifically grounded and validated models
CourseMap AI is built on mathematical foundations developed over more than a decade of research in AI and neuroscience. The underlying methods have been patented and published in leading peer-reviewed journals, and validated through large independent studies involving thousands of patients.
Models have been constructed and published for Alzheimer’s, Parkinson’s, Huntington’s disease, and Amyotrophic Lateral Sclerosis.
This scientific grounding ensures that the models are not only predictive, but stable, interpretable, and suitable for use in regulated clinical research environments.
Profiling trial participants for more precise treatment effect estimates
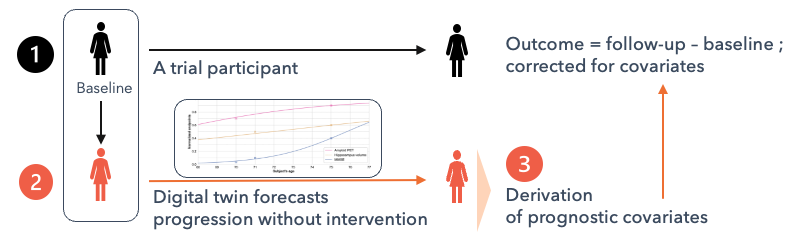
CourseMap AI creates digital twins of trial participant at baseline. These models forecast disease progression over time. From these forecast one may derive prognostic covariates identifying fast vs. slow, and early vs. late progressors. By accounting for inter-patient variability within the trial population, this approach reduces outcome variance, resulting in a tighter confidence interval of the outcome.
An approach applicable regardless of the mechanism of action
In neurodegenerative trials, treatments are designed to slow disease progression or delay symptom worsening. As a result, the magnitude of the observed treatment effect depends in part on the participant’s underlying progression rate at trial entry. Participants who are expected to progress rapidly are more likely to show a larger measurable treatment effect—assuming the treatment is effective—than participants whose disease is expected to progress slowly or remain stable over the same period.
Prognostic covariates capture this variability in disease progression and, which is therefore partly predictive of the treatment response. By accounting for these differences, prognostic covariates reduce unexplained variance in outcomes when treatments are evaluated in heterogeneous patient populations with diverse progression profiles. Importantly, this reasoning applies independently of the treatment’s mechanism of action.
Patented technology and validated models in several indications
Best forecast of cognitive decline due to Alzheimer's disease
An approach already deployed in pharmas and biotechs
Ready to boost your trial with AI?
Let’s discuss how Qairnel’s predictive models can help you optimize patient selection and reduce trial variability.
